Targeting Malignant Synapses to Prevent Glioblastoma Recurrence
Source PublicationCancer Discovery
Primary AuthorsDing, Dong, Pan et al.
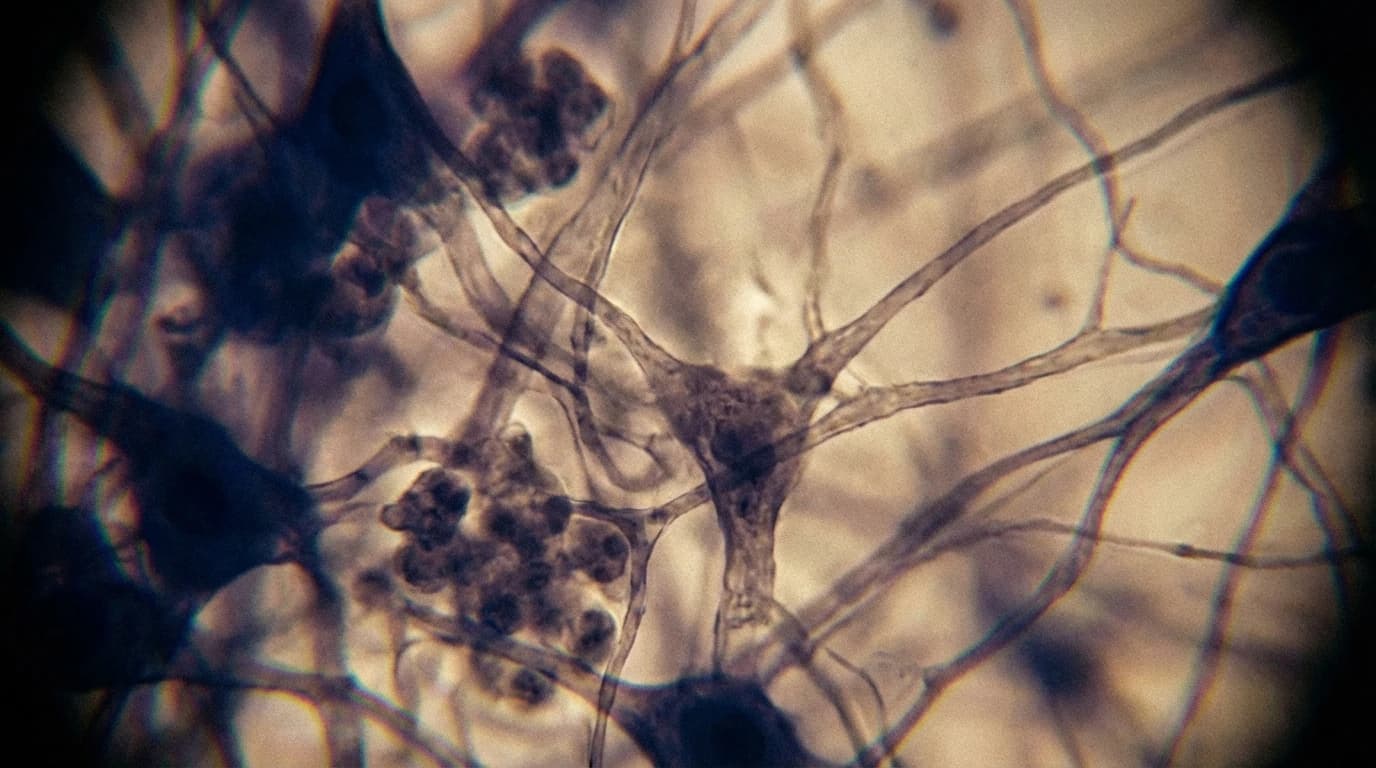
The Hidden Driver of Glioblastoma Recurrence
Researchers have successfully blocked brain tumour cells from physically wiring themselves into healthy neural circuits, a feat previously considered nearly impossible because the cancer cells embed so deeply into the host tissue. This insidious integration is the primary engine behind glioblastoma recurrence. Historically, oncology has viewed tumours as isolated, rapidly dividing masses that can be cut out or poisoned.
These results were observed under controlled laboratory conditions, so real-world performance may differ.
The old method relied entirely on surgical excision, radiation, and broad-spectrum chemotherapy. However, this approach fails because it ignores the structural reality of the disease. Glioblastoma cells extend long, thread-like structures called tumour microtubes that form active, functional synapses with healthy neurons. These malignant connections feed the cancer, allowing it to survive aggressive treatments and return with devastating speed.
Identifying the Chemical Messenger
The researchers measured the specific signalling molecules exchanged between the tumour and the brain. They found that glioma cells secrete a protein called C1QL1. This protein binds to a specific receptor, BAI3, located on both neighbouring healthy neurons and other cancer cells.
Once bound, it triggers a protein called Rac1, which forces the healthy neurons to prune their normal connections. To test a solution, the scientists applied a novel Rac1 inhibitor. The laboratory results showed that this compound successfully rescued healthy synapses and stopped the tumour from building new microtubes.
Clinical Implications and Future Hurdles
By targeting the physical communication between cancer cells and neurons, this intervention suggests a highly targeted way to manage the disease. If the tumour cannot build its network, it may remain a distinct, vulnerable mass. The immediate benefits observed in the laboratory include:
- Prevention of tumour microtube expansion into healthy tissue.
- Protection of normal synapses from tumour-induced pruning.
- Disruption of the overarching glioma network.
What the Study Does Not Solve
Despite the elegance of this biological mechanism, the study leaves several massive hurdles uncleared. Most notably, it does not solve the problem of drug delivery to the central nervous system. The blood-brain barrier blocks the vast majority of therapeutic compounds, and researchers have yet to demonstrate that this specific Rac1 inhibitor can safely reach effective concentrations in human patients without severe toxicity.
Furthermore, cancer biology is highly adaptive. It remains entirely unknown whether the tumour might bypass the C1QL1 pathway and use an alternative method to build its microtubes once the primary route is blocked. Moving from cell cultures to human trials will require extensive safety profiling to ensure that inhibiting Rac1 does not also degrade healthy cognitive functions.