Genetics & Molecular Biology1 March 2026
How a Common Genetic Switch Could Advance von Willebrand Disease Gene Therapy
Source PublicationBlood Advances
Primary AuthorsBär, Groten, Barraclough et al.
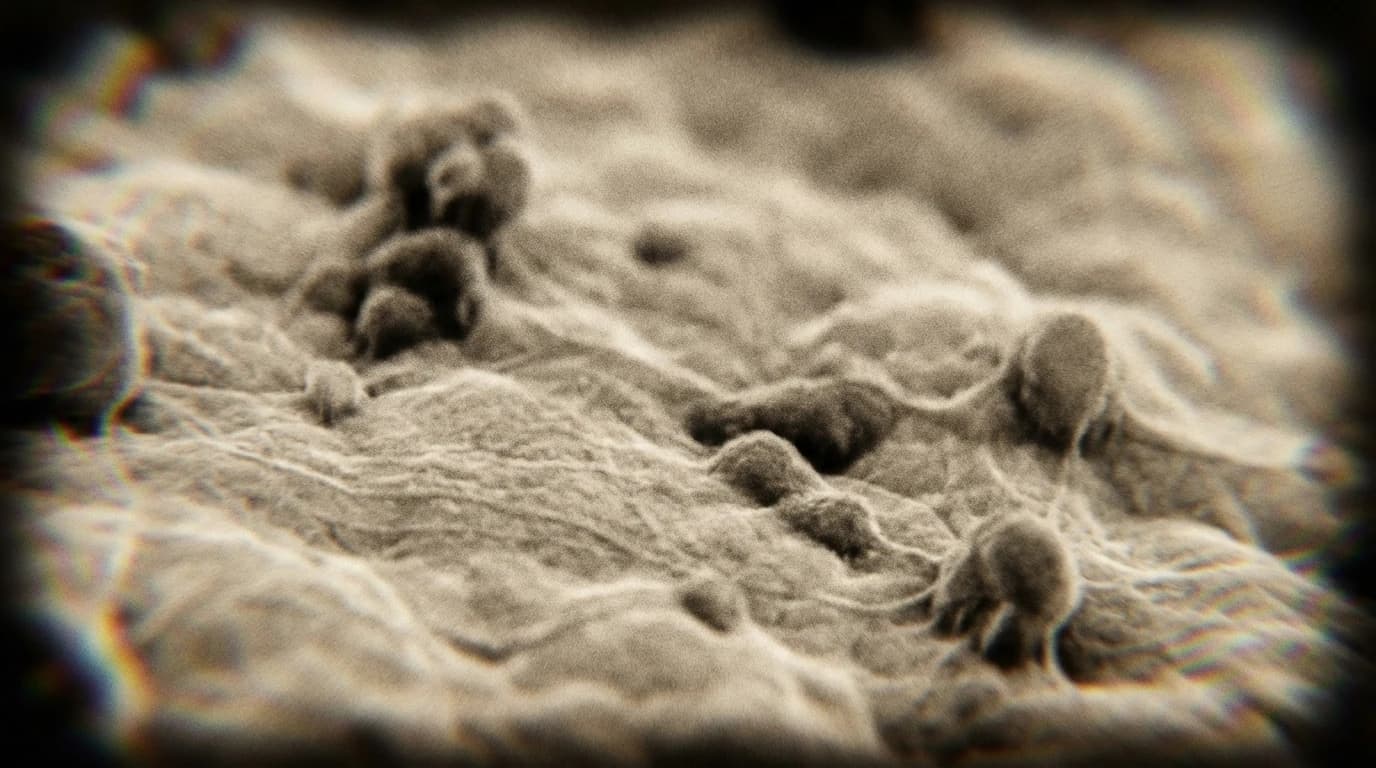
These results were observed under controlled laboratory conditions, so real-world performance may differ.
Von Willebrand disease (VWD) is a common bleeding disorder caused by a missing or defective clotting protein. In severe cases, patients inherit one healthy gene and one faulty, dominant gene that disrupts the entire clotting process. Creating a specific therapy for every possible mutation is practically impossible. Researchers needed a way to target the faulty gene without having to design a custom treatment for each individual patient.
Instead of chasing rare mutations, scientists targeted a common genetic variation known as a single nucleotide polymorphism (SNP). Using CRISPR/Cas9, they selectively disabled the faulty allele in endothelial cells taken from patients with VWD. The study measured efficient, allele-selective knockout of the defective protein.
Mass spectrometry confirmed that the healthy protein continued to express normally. Furthermore, the researchers observed a reversal of the disease phenotype in these lab-grown cells. By focusing on a shared genetic trait rather than the specific disease-causing flaw, the team bypassed the need for highly individualised medicine.
The Future of von Willebrand Disease Gene Therapy
This approach changes the trajectory of genetic medicine for bleeding disorders over the next decade. By targeting a common genetic marker rather than individual mutations, a single therapy could treat a wide swathe of patients. This drastically reduces the time and cost associated with drug development.We are moving away from an era where rare, heterogeneous mutations prevent the development of viable treatments. Over the next five to ten years, this strategy suggests a shift away from lifelong infusions. If these ex vivo results translate to human trials, the downstream applications could include:
- One-time genetic interventions replacing regular intravenous therapies.
- A single, broadly applicable treatment for multiple VWD subtypes.
- Adaptation of this technique for other dominant genetic disorders.
Future clinical trials will need to confirm safety and delivery methods in living patients. Targeting endothelial cells inside the human body remains a complex logistical hurdle. However, the data suggests that a scalable approach to managing complex genetic diseases is well within our reach.
Cite this Article (Harvard Style)
Bär et al. (2026). 'Allele-selective disruption of pathogenic VWF variants in type 2 von Willebrand disease using CRISPR/Cas9.'. Blood Advances. Available at: https://doi.org/10.1182/bloodadvances.2025018760