Guar gene editing: How scientists cracked the tough exterior of a climate-resilient crop
Source PublicationThe Plant Genome
Primary AuthorsGhosh, Biswas, Malaker et al.
The Brick Building and the Recipe
Imagine you are trying to rewrite a secret recipe locked inside a heavy steel safe. This safe is hidden inside a thick, heavily fortified brick building.
These results were observed under controlled laboratory conditions, so real-world performance may differ.
To edit that recipe, you cannot just walk through the front door. You first have to carefully dissolve the brick walls without damaging the safe inside.
Plant cells are remarkably similar to that brick building. They possess tough, rigid cell walls that protect them but make them notoriously difficult for scientists to alter.
Why Guar Gene Editing Matters Now
Guar is a highly durable, climate-resilient legume. We increasingly rely on it as a valuable commodity for a massive range of industrial and agricultural applications.
As global weather patterns shift, agricultural systems need crops to be even hardier. This is where Guar gene editing enters the picture.
Despite the high commercial value of the crop, scientists had never successfully modified the DNA of this specific plant. The thick cellular walls continually blocked modern genetic tools like CRISPR from getting inside.
Dissolving the Walls
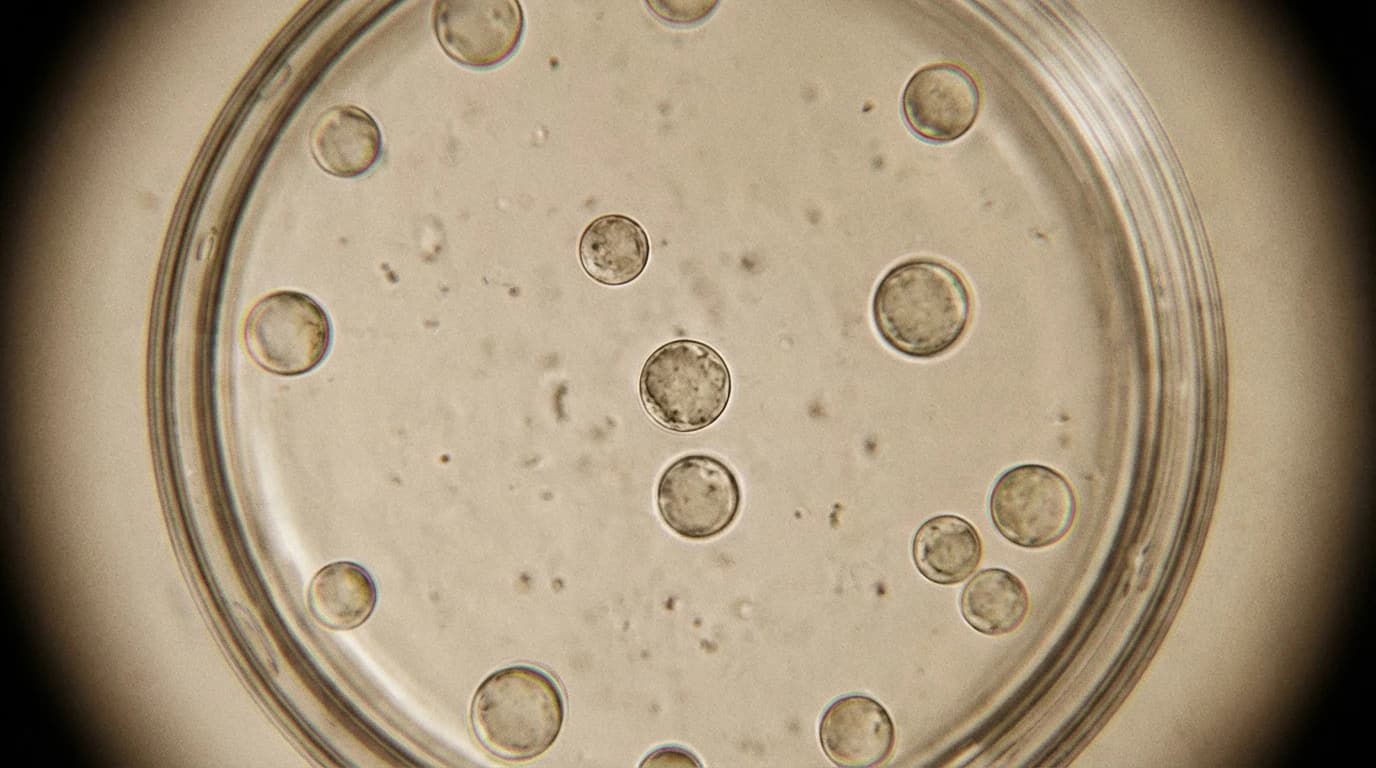
Researchers have finally bypassed these defences, publishing the first successful protocol for Guar gene editing. They began their experiment with six-day-old guar seedlings.
Using a clever 'tape-sandwich' technique, they physically peeled away the outer plant layers. Following this, they soaked the plant tissue in a precise mixture of enzymes, including cellulase and pectinase.
These enzymes acted like chemical wrecking balls, digesting the tough cell walls. This process left the researchers with 'protoplasts', which are naked plant cells completely stripped of their brick-like exterior.
Delivering the Genetic Scissors
With the outer walls gone, the safe was finally exposed. The researchers then used a chemical called PEG to slip the CRISPR machinery straight into these vulnerable cells.
They programmed their CRISPR tools to target a specific sequence known as the CtPDS gene. The goal was to see if the genetic scissors could accurately find and cut this exact location.
What This Means for Agriculture
The lab tests measured a flawless 100 per cent editing efficiency in the samples that successfully absorbed the CRISPR tools. The team recorded large fragment deletions in the target gene, confirming the system worked exactly as intended.
While this study relies on a transient system—measuring effects on isolated, wall-less cells in a lab rather than fully grown plants—the findings establish a reliable baseline. Building on this protocol may allow researchers to design crops with highly desirable traits.
Future applications of this technique could include:
- Unlocking functional genomics to better understand the plant's underlying biology.
- Enabling targeted trait improvement for shifting agricultural needs.
- Driving general crop improvement to maximise guar's industrial value.