Cracking the Castle Wall: The First Success in Guar Genome Editing
Source PublicationThe Plant Genome
Primary AuthorsGhosh, Biswas, Malaker et al.
The Medieval Castles of Plant Biology
Imagine a plant cell as a tiny medieval castle. It possesses a rigid outer wall built specifically to keep microscopic invaders out. If you want to rewrite the royal decrees—the DNA—hidden deep inside the keep, you cannot just knock on the front door.
These results were observed under controlled laboratory conditions, so real-world performance may differ.
You have to temporarily dissolve that heavy stone wall without destroying the delicate castle itself. For years, this formidable barrier has frustrated biologists working with guar, a tough legume prized for its industrial and agricultural uses.
Why Guar Genome Editing Matters Now
Guar is a remarkably climate-resilient plant with a rapidly growing list of commercial applications.
Despite its increasing value as a commodity, scientists have struggled to update its genetics. Guar genome editing has remained an elusive target because getting genetic instructions past the plant's tough exterior is notoriously difficult.
To build a foundation for future crop improvement, researchers needed a reliable way to edit its DNA. Now, they have finally found a way to breach the walls.
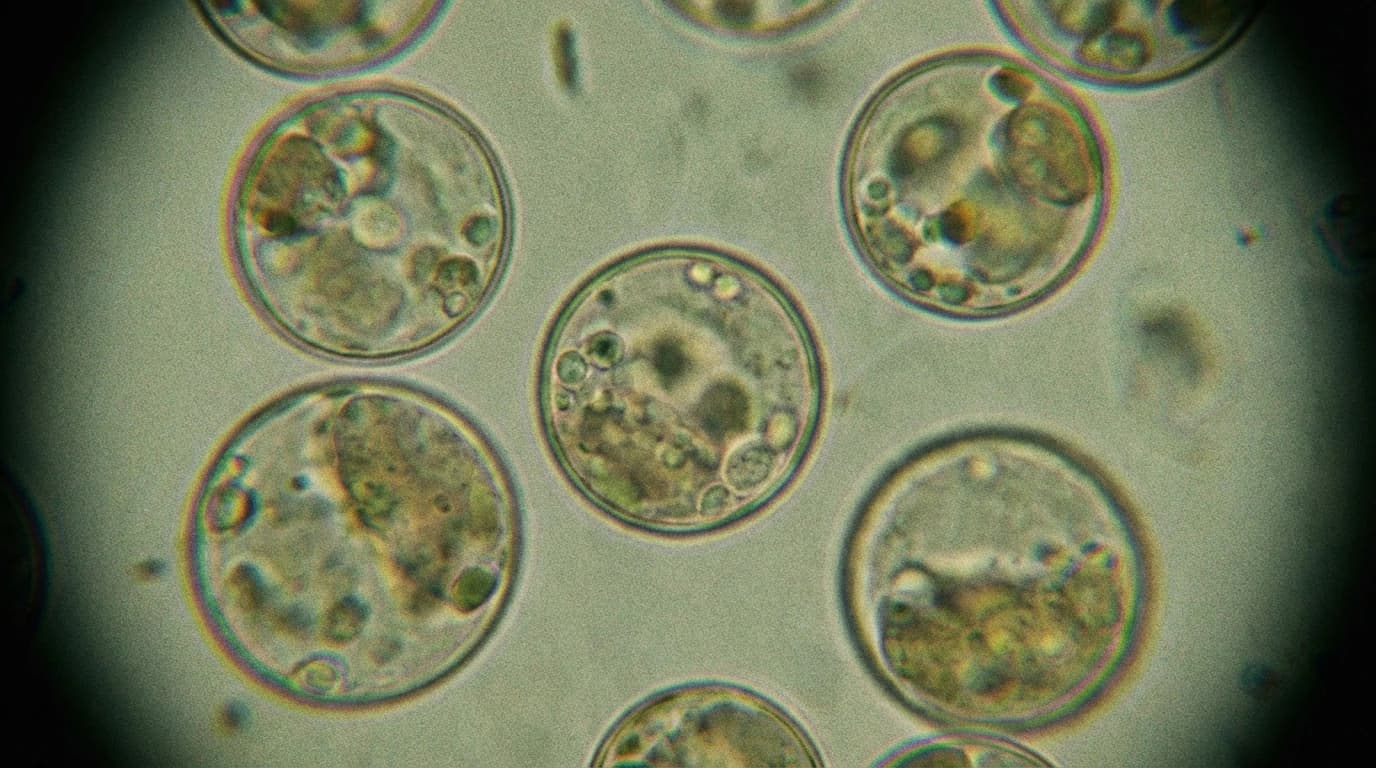
How to Breach a Cellular Fortress
In a recent lab study, scientists developed a precise chemical siege to slip past the plant's defences. They focused on creating naked, wall-less plant cells known as protoplasts.
The researchers broke the process down into three distinct steps:
- First, they applied a clever 'tape-sandwich' method to the cotyledons (the embryonic seed leaves) of six-day-old seedlings, physically peeling away the outermost protective layers.
- Next, they soaked the exposed tissue in a liquid cocktail of enzymes, which safely digested the rigid cellulose walls.
- Finally, they used a chemical compound called PEG to temporarily open pores in the inner membrane, allowing CRISPR/Cas9 genetic scissors to slip inside.
Once inside, the CRISPR machinery was programmed to find and cut a specific target called the CtPDS gene.
Measuring the Impact
The precision of this microscopic siege was remarkable. When the researchers sequenced the DNA of the transfected protoplasts, they measured a 100 percent editing efficiency in the successfully altered cells.
Every single cell that took up the CRISPR tools showed the exact genetic deletions the team intended to make. The resulting DNA cuts ranged from 714 to 1061 base pairs long, proving that the biological scissors function perfectly once they actually get inside the guar cell.
While currently demonstrated at the laboratory bench as a transient system for functional genomics, this optimised protocol provides a crucial first step for agricultural engineering. By proving that targeted edits are practically achievable in these isolated cells, scientists now have a reliable foundation for future crop improvement, opening the door to targeted trait enhancements down the line.